
VHH Antibodies
Specifica delivers VHH antibody libraries and discovery campaigns with the same expertise we apply to our ScFv and Fab libraries.
Read about our success in developing single-domain (VHH) antibodies from our libraries in this June 2025 article in mAbs.
Recombinant VHH libraries from Specifica deliver:
- Fully humanized VHHs – not just the frameworks, but also the CDRs are humanized, providing improved safety and therapeutic efficacy
- Broad diversity – our phage display libraries far exceed the diversity of immunization, and sorting via yeast display provides additional capabilities to enable selection for desired properties
- High specificity – controlled selection conditions allow you to focus on specific regions of your target antigen
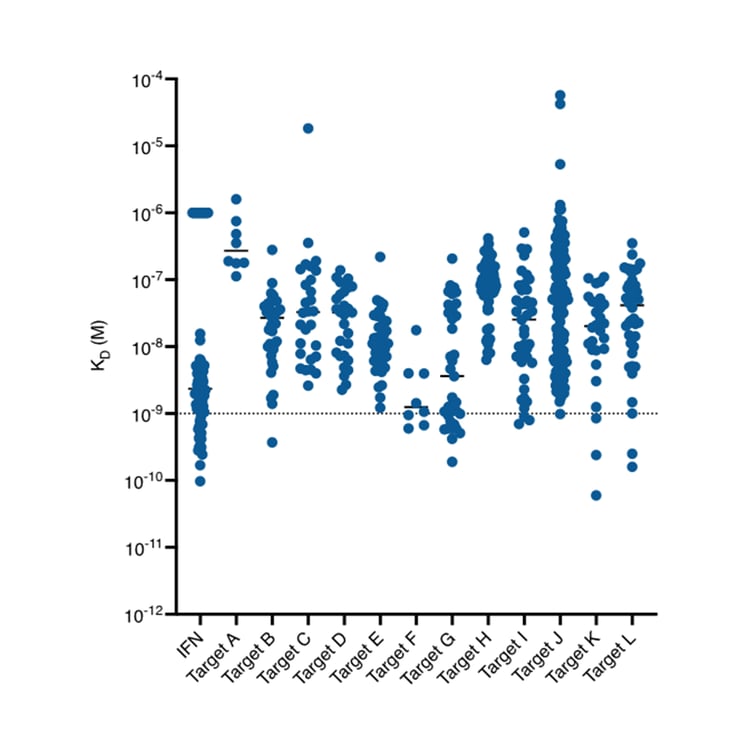
- High affinity – exceptional binding due to the preserved CDR regions, often reaching into the sub-nanomolar range
- >50% of campaigns yield at least one subnanomolar binder
- >50% of binders <10nM and ~5% of binders are subnanomolar
- High affinity and specificity – combined, high affinity and desired specificity can provide a drug-like VHH straight from selection, often without the need for additional improvement
- High developability – over 70% of VHHs have two or fewer biophysical liabilities
Together, our proven scaffolds, desired specificity, high affinity and excellent developability can provide a drug-like VHH straight from selection, often without the need for additional improvements.
What are VHH antibodies and what are their advantages?
- VHHs are single-domain antibodies based on camelid antibodies
- VHHs are much smaller than traditional antibodies, enabling them to bind to challenging targets including “hidden” epitopes, less easily accessible by traditional antibodies
- VHHs are generally more diverse than other antibody fragments, due to their unique structure, with elongated CDR1 and CRD3 loops
- VHHs are stable and soluble, making them well suited to meet the developability requirements of consistent quality and reduced variability
What is Specifica's approach to building VHH libraries?
- Our VHH libraries are based on four well-behaved therapeutic VHH molecules, with HCDR1 and 2 natural replicated diversity from rearranged VH3 family genes;
- The CDRs are filtered for correct folding via protein A selection (protein A binding being an inherent property of the VH3-23 gene family)
- Only well-folded CDRs are allowed in the final library, enhancing its functionality and overall quality.


